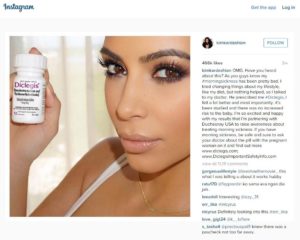
So. Kim Kardashian. I hate to even post her name because all it does is strengthen her multi-million dollar empire of nothingness. And part of that nothingness is the posting of countless product placement bits on social media like Instagram. You may think it’s a harmless conduit between brainless posters and brainless viewers, but what it really represents (at least for super popular accounts like Kardashian’s) is a vast advertising medium. Yes, advertisers pay those popular posters to showcase their products.
“OMG. Have you heard about this?” Kim posted, along with a picture of her prescription morning sickness drug. But this wasn’t just an innocent post from an end-product user touting her happiness with the item. She was paid to post this, and the brief post went to her 70 million viewers–without the FDA oversight required for direct advertising.
Yes, the FDA strictly regulates the advertising of drugs. If you haven’t paid this any attention, simply look at the number and type of disclaimers that each televised pharmaceutical ad contains. These are not voluntary…who wants to hear about diarrhea, incontinence, or death when you are hearing about something as fun as the commercials otherwise make the drugs seem? One minute you’re dancing to Walking on Sunshine and then there’s that pesky erection lasting longer than 4 hours “problem”. Let’s face it, the FDA is a no-nonsense, no sense-of-humor governmental agency tasked with protecting the American health product consumer from the multi-billion dollar pharmaceutical industry’s influential ways.
The vast new horizon of social media advertising opens some tricky questions. Does the FDA have oversight over unofficial advertising media such as Facebook, Instagram, or even a blog? If there is not a law broken, is it still a moral or ethical concern? Does direct advertising affect consumer requests for drugs, and physician prescribing patterns? What do you think?
I guess I’ll just finish by saying I have no financial interest in this product or any competitor. You know, in case the FDA asks.






People who follow her deserve what they get. We need to thin the heards anyway